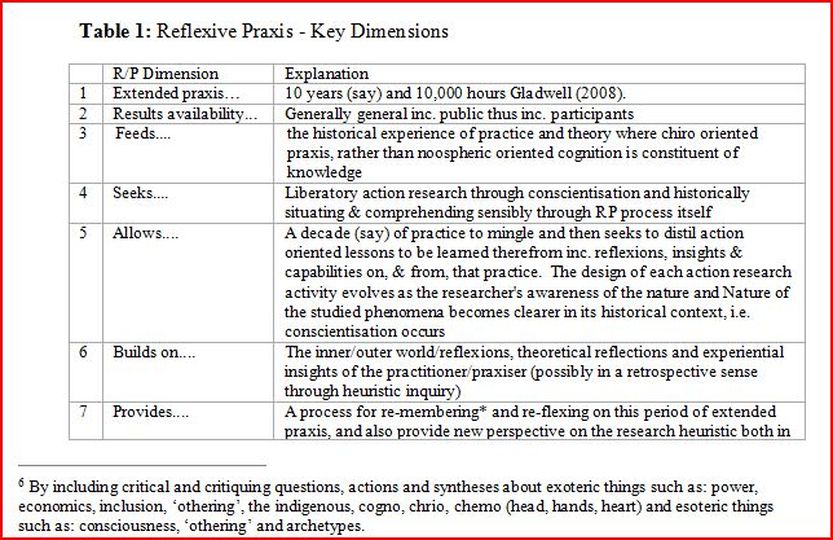
Examples - Consent and Participant information sheet.
Participant Information Sheet. 1. Research Project Title. Research data management and libraries international survey. 2. Invitation. You are being invited to take part in this research project. Before you decide to do so, it is important you understand why the research is being done and what it will involve.PARTICIPANT INFORMATION SHEET TAKING PART IN AN INTERVIEW - A STUDY OF WOMEN’S EXPERIENCES OF DOMESTIC VIOLENCE AND ABUSE Hello Our names are Maggie Evans and Emma Johnson, researchers from Bristol University. We are asking you to take part in research. Before you decide if you want to take part or not.The Participant Information Sheet (PIS) should be a clear and simple document, on headed paper with the University Crest (or equivalent for other institutions), that would be easily understood by those to whom it is aimed; for example, it should be age-appropriate.
This example shows how Flesch Reading Ease score or Fog score can be calculated. This helps improve readability of your Participant Information Sheet and Consent form. Count the words and sentences. Divide the number of words by the number of sentences. Count the long words (more than two syllables).Participant Information Sheet The title of the research project. Mechanisms of tactile signal detection You are being invited to take part in a research study. Before you decide whether or not you want to take part it is important for you to understand why the research is being done and what it will involve.
Preparing a participant information sheet You must give any potential participants in a research project enough information for them to make an informed decision about whether they want to take part. A recognised approach is to provide potential research participants with an information sheet that explains the purpose of the research and the.